 
Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (Cl 2) Count i Weight i. Then, lookup atomic weights for each element in periodic table: Cl: 35.453. The molar mass of a compound is the mass in grams of 1 mole of that substance, a property expressed in units of grams per mole (g/mol)īecause the definitions of both the mole and the formula mass are based on the same reference substance, 12C, the molar mass of any compound is numerically equivalent to its formula weight in amu. First, compute the number of each atom in Cl 2: Cl: 2. The masses of 1 mole of different compunds, however, are different, since the masses of the individual molecules are drastically different. Similar to atoms, 1 mole of any compound contains the same number of molecules as 1 mole of any other compund. Let’s walk step-by-step for finding the molar mass of calcium chloride (CaCl2): First, write out the chemical formula for the compound. 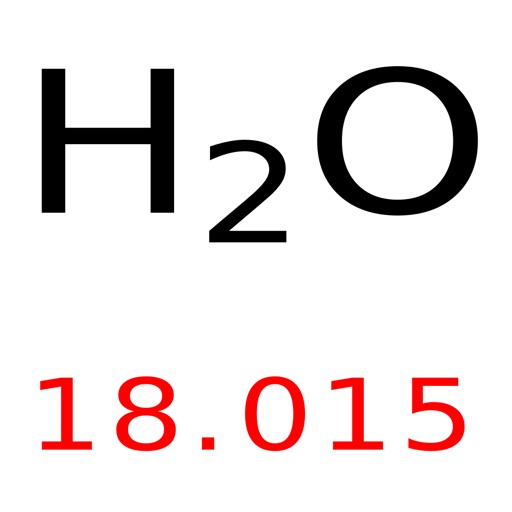
Plus, often, in exams, atomic or molar masses are provided, so you would use the rounding provided.\). My TA also advised us to round at the very end if we can, and use the same number of significant figures as the values given in the question. 
The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6.022 × 10 23 carbon atomsis therefore 12.011 g/mol: Table 2.4.1 2.4. It can be created by neutralising hydrochloric acid with calcium hydroxide. I'll also add for your specific question regarding exams that my TA told me our midterm and final exams will have an online format similar to the Achieve HW, so if you are finding the rounding of answers on there fine you should be ok on the exams as well. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. Calcium chloride is an inorganic compound, a salt with the chemical formula CaCl 2.It is a white crystalline solid at room temperature, and it is highly soluble in water. 42.5-42.9), I would use the longer versions of the atomic masses and round your answer at the end to the same number of significant figures as the value given to you in the question (or by using the general rounding rule of rounding to the same number of significant figures as the least precise value - so the value with the least number of significant figures - in the calculation), as it ensures you don't lose accuracy in the calculations and end up with the correct answer. However, for questions with calculations of mols, concentrations etc, where the answer can be a decimal which has to be precise to a certain number of significant figures (especially important if you are unsure whether the question you are working on has a range of acceptable answers e.g. If you are calculating an empirical formula, for instance, you are looking for a whole number, so using the rounded atomic masses would be fine as it would give you a number that, even if slightly different from the number you would get by using the atomic numbers "in full", would still round to the same whole number and ultimately give you the right answer. I think it often depends on the type of question. The periodic table lists the atomic mass of carbon as 12.011 amu. Then, lookup atomic weights for each element in periodic table: Ca: 40.078, Cl: 35. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. Computing molar mass step by step First, compute the number of each atom in CaCl 2: Ca: 1, Cl: 2. Hi, I'll copy below the answer I gave to someone who had a very similar question to yours. In each case, the number of grams in 1 mol is the same as the number of atomic mass units that describe the atomic mass, the molecular mass, or the formula mass, respectively. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
